Amphoteric surfactant-type antimicrobial agent"LEBON T-2"

By blending with amphoteric or nonionic surfactants, the sanitizing detergent can be adjusted to combine antibacterial and cleaning power.
What's great about SANYO CHEMICAL's amphoteric surfactant-type antimicrobial agents "LEBON T-2"
Amphoteric surfactant type antimicrobial agent "LEBON T-2"
By blending with amphoteric surfactants and non-ionic surfactants, a sanitizing detergent with both antimicrobial and cleaning power can be adjusted.
・ Exhibits a broad antimicrobial spectrum.
・ Demonstrates stable antimicrobial activity, with little precipitation or decrease in antimicrobial activity due to organic matter such as proteins.
・Demonstrates excellent antimicrobial activity even in alkaline conditions.
Structural formula of active ingredient
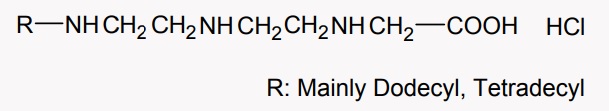
Appearance: Pale yellow liquid (may turn cloudy and solidify at low temperatures)
Odor : Slight peculiar odor
Active ingredient : Approx. 40% by mass
pH : Approx. 7 (2.5% by mass in water)
Examples of Uses
The following are examples of formulations of sanitizing detergents for use in food processing plants.

Example of formulation (indicated by concentration of active ingredients)
(A)LEBON T-2 : 4 mass%
(B)Nonionic surfactant : 20 mass%
(C)Fatty acid ethanolamide : 5 mass%
(D)EDTA : 1 mass%
(E)Sodium hydroxide: Appropriate amount (amount to bring pH to 9)
Water : Remaining amount
-----------------------------------------------------------------------
Total 100
Example of blending method
While stirring water, add (B) and dissolve uniformly. Then add (A), (C), (D), and (E), and stir until uniform. (No heating or cooling is required.)
Performance data
Minimum inhibitory concentration (MIC) of "LEBON T-2" against bacteria and fungi
Minimum inhibitory concentration (MIC) of "LEBON T-2" against various bacteria
| bacterial strain | MIC mass ppm (active ingredient) |
|---|---|
| Pseudomonas aeruginosa | 400 |
| Pseudomonas cepacia | 200 |
| Escherichia coli | 100 |
| Bacillus subtilis | 50 |
| Staphylococcus aureus | 12.5 |
Minimum inhibitory concentration (MIC) of "LEBON T-2" against various fungi
| Bacterial strain | MIC mass ppm (active ingredient) |
|---|---|
| Penicillium citrinum (blue mold) | 800 |
| Rhizopus oryzae | 400 |
| Aspergillus niger (black mold) | 400 |
Test Method
Results of drug dilution agar plate method according to the MIC measurement method of the Japanese Society of Chemotherapy.
Medium: Mueller Hinton medium (for bacteria)
Potato dextrose agar (for fungi)
Inoculum: Approximately 106 CFU/mL
Reference
Yamamoto, N., Doi, J., Takada, K., Hashimoto, I., Shinoda, K., and Oyama, A., Journal of Bacteria and Mold Control, 18(10), 477-483 (1990).
Usage
For use as-is with only dilution
Depending on the purpose and conditions of use, dilute the product with tap water so that the concentration of the active ingredients ranges from several mass ppm to several thousand mass ppm.
When blending with other agents to produce sanitizing detergents, etc.
Please check the stability and antimicrobial activity with other ingredients before use.
When used at low temperatures
Turbidity may occur at low temperatures. If turbidity occurs, heat the product to 50-60°C to make it homogeneous before use.
Precautions against misuse
When mixed with other drugs, precipitation may occur depending on the temperature and concentration used. When using this product in combination with other ingredients, check the stability of the mixture beforehand and use it within the range where there are no problems.
Examples of substances that are likely to cause precipitation include the following
Iodine tincture, phenol, sulfosalicylic acid, hydrogen peroxide, silver nitrate, tannic acid, sodium sulfosalicylate, potassium permanganate, potassium dichromate, etc.
Please be sure to read the "Safety Data Sheet" (SDS) for each product and sub-material (chemical) before handling the products listed on this website. The SDS can be obtained from here. https://www.sanyo-chemical.co.jp/products/en/sds/
Related Information
Related Products
-
Link to Sanyo Chemical's corporate website
This page has been prepared solely for information purposes.
Sanyo Chemical Industries, Ltd. extends no warranties and makes no representations as to the accuracy or completeness of the information contained herein, and assumes no responsibility regarding the suitability of this information for any intended purposes or for any consequences of using this information.
Any product information in this brochure is without obligation and commitment, and is subject to change at any time without prior notice.
Consequently anyone acting on information contained in this brochure does so entirely at his/her own risk.In particular, final determination of suitability of any material described in this brochure, including patent liability for intended applications, is the sole responsibility of the user. Such materials may present unknown health hazards and should be used with caution. Although certain hazards may be described in this brochure, Sanyo Chemical Industries, Ltd. cannot guarantee that these are the only hazards that exist.





